Despite its status as a rare disease, amyotrophic lateral sclerosis (ALS) has been in the public consciousness for over a century. One of the most conspicuous symbols of ALS that persists is New York Yankee, Lou Gehrig, who announced that he was afflicted with the disease over 80 years ago. He died two years after his statement and the world mourned one of baseball’s most admired figures. Since then, ALS has maintained a place in the forefront of the public’s mind and is frequently referred to as Lou Gehrig’s disease. Another prominent and inspiring figure with ALS was renowned physicist Stephen Hawking. He commanded the scientific world’s attention for decades, with widely read books on black holes and the big bang and was the subject of a popular 2014 movie. Also in 2014, the Ice Bucket Challenge raised awareness and funds for ALS research with politicians, actors and other well-known figures playing along and contributing to the lighthearted but effective fundraiser.
The attention directed towards ALS has been successful and led to growing resources for ALS research. Most recently, the FDA granted approval to Relyvrio on September 29, 2022, the first since 2017. There are other treatments for ALS available, including Riluzole and Radicava that focus on functional change in the patient. However, there remains a significant unmet need for both functional and survival benefit improvements that may be achievable by new, in-development candidates.
ALS and other neurodegenerative diseases share a common underlying mechanism: a decline in the brain cells’ ability to produce energy. Although this decline occurs as a part of natural aging, it is amplified and exacerbated by neurodegenerative disease. Related energy deficits result from and lead to neuronal dysfunction and death. If science can address this, we could drive critical cellular energy-producing reactions in the brain to treat neuronal energetic failure and produce disease-modifying effects in ALS.
Clene Inc. (NASDAQ: CLNN) is one of the leaders in the ALS space, with a clinical stage, neurodegenerative therapy-focused pipeline offering programs in ALS, Parkinson’s disease (PD) and Multiple Sclerosis (MS). Its neurodegenerative indications rely on CNM-Au8, a gold nanocrystal suspension that has demonstrated a survival benefit in multiple ALS trials. The lead program in ALS recently completed the HEALEY ALS Platform Trial. While the study with CNM-Au8 did not achieve its primary endpoint, it did show a reduction in risk of death and permanently assisted ventilation at 24 weeks.
Clene Pipeline[1]

CNM-Au8
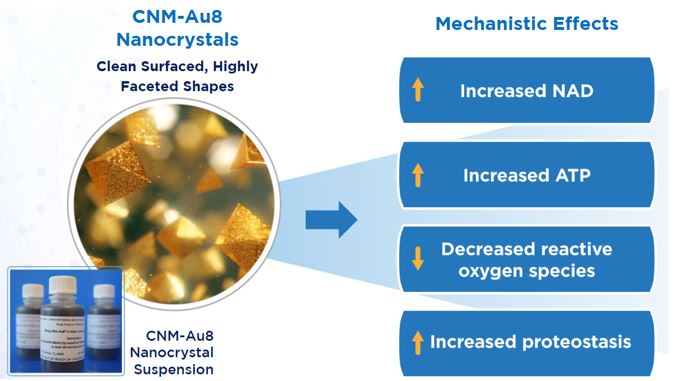
The drug at the center of Clene’s lead programs is CNM-Au8. CNM-Au8 is a drinkable, catalytically active gold nanocrystal suspension that can cross the blood-brain barrier (BBB) and is designed to improve cellular energy production and utilization that enables neuroprotection and remyelination. The faceted crystals are ~13 nm in diameter with surfaces that catalyze oxidation-reduction reactions, including the conversion of NADH[2] to NAD+[3],[4] a process that is key to converting food into energy. NAD+ is an essential component of cellular energy and a cell metabolic sensor. The catalytically active nanocrystals of CNM-Au8 stimulate cellular energy production that protects the brain and the protective coating around nerve fibers by increasing resilience to environmental stressors. Gold nanocrystals may provide a therapeutic benefit by correcting cellular-energy deficits and relieve the oxidative stress present in multiple neurodegenerative diseases.
CNM-Au8 Nanocrystals[5]

Healey ALS Platform Trial
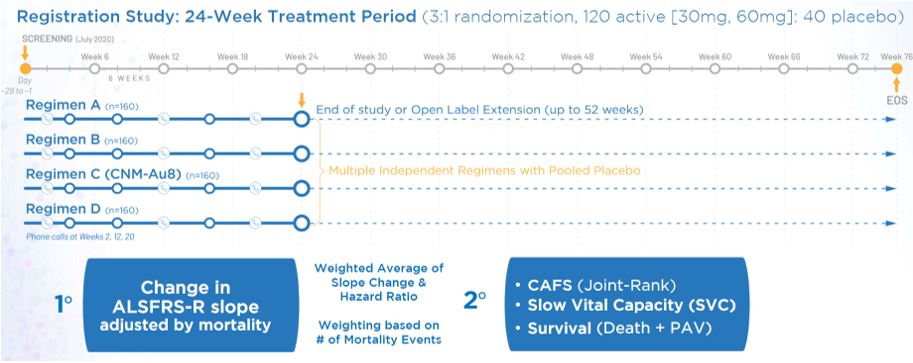
Following a Phase I safety trial in healthy volunteers and the Phase II RESCUE-ALS and REPAIR trials demonstrating target engagement in the brain, Clene was selected to participate in the Healey ALS Platform Trial (Healey) in May 2020 along with several other sponsors.[6] The Healey trial is a multicenter, double-blind, placebo controlled, perpetual adaptive platform trial which is investigating multiple candidates in parallel for ALS. It shares a control arm with the other therapies, which increases the likelihood that study participants will receive the investigational drug, shortens the length of the trial and reduces cost. The trial is run by the Sean M. Healey and AMG Center for ALS at Mass General in Boston, Massachusetts with active sites throughout the United States.
Healey ALS Platform Trial Design[7]

The CNM-Au8 arm of the Healey trial enrolled 160 subjects with 120 administered CNM-Au8 and 40 placebo.[8] The primary endpoint was change in disease severity over time as measured by the slope of change in the ALS Functional Rating Scale-Revised (ALSFRS-R). Secondary measures included combined assessment of function and survival (CAFS), respiratory function measured by slow vital capacity (SVC) and survival measured by death or use of permanent assisted ventilation (PAV).
CNM-Au8 Healey Trial Results
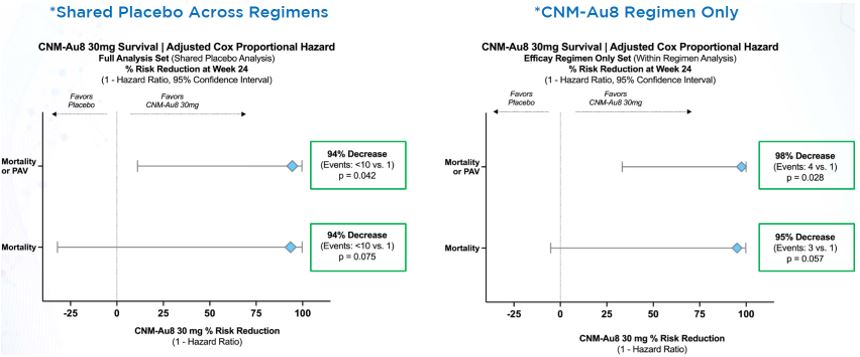
In October 2022, Clene reported topline results for the Healey trial in a press release and investor call. CNM-Au8 did not achieve the primary endpoint for ALSFRS-R at 24 weeks and did not show significant change in CAFS and SVC. However, there was a greater than 90% risk reduction in survival as measured by mortality and use of PAV for the 30 mg dose. This favorable result was also supported by the data generated in the 2020 RESCUE-ALS trial which demonstrated a 70% decreased risk of death for participants in the CNM-Au8 arm.
Survival Benefit for CNM-Au8[9]

Clene is gathering additional long-term data which, if supportive, may provide additional confidence around survival endpoints. Biomarker data is being collected from urine, plasma and blood to help make the regulatory case. An end of Phase II meeting is expected mid-2023 which will provide the pathway forward which may include submission for accelerated approval and a Phase III confirmatory study.
Safety
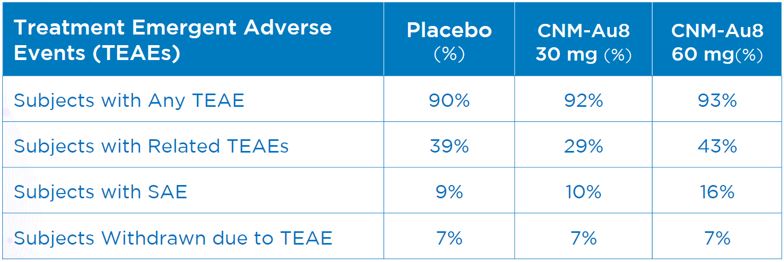
Safety was a strong point for CNM-Au8 which was well tolerated and produced no definitive safety signals. Treatment emergent adverse events (TEAEs) were similar between the 30 mg CNM-Au8 and placebo arm and there were no serious adverse events (SAEs) associated with CNM-Au8. As can be seen in the following summary of topline safety data, TEAEs and SAEs were higher in the 60 mg group as compared to the 30 mg group. 30 mg will be used in future pivotal studies. Additional analysis of data generated by the trial will be conducted over the coming months to provide greater insight into the results and genomic and biomarker data will be evaluated.
Summary of Safety Data from Healey Trial[9]

Biomarkers
Biomarkers serve a valuable role providing early evidence of efficacy in diseases that can take years to manifest. They are biological molecules or genes that can be used as indicators of a specific physiological or pathological process in the body which can be used to diagnose a disease, predict its progression, monitor treatment effectiveness, and identify a person's risk of developing a particular condition. This is especially important in ALS where it takes about a year to diagnose after first symptoms and early treatment may have the most impact. Several neuroinflammatory biomarkers have shown a correlation with ALS disease process and will be evaluated by Clene. These include neurofilament light chain (NfL)[10], p75[11] and UCHL1[12].
Next Steps
It is important to note that Healey was a Phase II trial and its goal was to identify an effective dose, establish safety, and inform design for the registrational confirmatory trial, such as duration of treatment and measures of efficacy. Clene’s Chief Medical Officer noted that CNM-Au8’s mechanism of enabling energy metabolism and efficiency may not be reflected in the slope of ALSFRS-R change after only 24 weeks of treatment. Longer treatment duration may be appropriate for follow on studies.
Clear cut positive results are hard to come by in the neurodegenerative space, as evidenced by the many failures that have been suffered, especially in Alzheimer’s disease and in ALS. However, the unmet need is immense and it is important to help patients with existing therapies as the science is refined.
Given the difficulty in achieving recognized endpoints in ALS trials, we’ve seen a willingness from FDA to be flexible and to work with drug sponsors to find a pathway forward. Although ALSFRS-R is an important endpoint which attempts to summarize a variety of symptoms into quantitative scores that reflect overall function, patients may value other outcomes.[13]
There are precedents for the FDA encouraging sponsors to proceed despite missed endpoints. Safety for all of the most recent ALS candidates has been favorable, shifting regulators’ attention towards efficacy. However, there has been difficulty identifying suitable primary and secondary endpoints that capture the ideal patient benefits. Let’s take a look at some of the other candidates that are moving forward and the regulatory support they have received.
Other Advancing ALS Candidates
BrainStorm Cell Therapeutics (NASDAQ: BCLI) is developing a cell therapy designated NurOwn that completed a Phase III clinical trial to treat ALS. The trial did not meet statistical significance for its primary endpoint of ALSFRS-R but did show an increase in neurotrophic factors and reduction in neurodegenerative and neuroinflammatory biomarkers. Despite the miss, BrainStorm filed a biologics license application (BLA) for NurOwn and was issued a refusal to file (RTF) letter by the FDA which declined to accept the BLA. Patients did show beneficial clinical effects and exhibited favorable changes in relevant ALS-related biomarkers providing the impetus for BrainStorm to take advantage of the FDA’s offer for a Type A meeting. The meeting, which was scheduled for January 11, 2023, will indicate whether or not there is a pathway towards an FDA Advisory Committee (AdCom) meeting and potential approval.
Biogen’s (NASDAQ: BIIB) tofersen was the subject of a Phase III trial investigating the safety and efficacy of the candidate in ALS patients exhibiting a superoxide dismutase 1 (SOD1) mutation. Tofersen is an antisense oligonucleotide that also missed its primary endpoint of ALSFRS-R. In October 2021, Biogen announced topline results from the study, reporting that while it missed the primary endpoint, it did show evidence of benefit across other secondary and exploratory measures of biologic activity and clinical function. The FDA ultimately accepted Biogen’s NDA for tofersen based upon improvements in the biomarker NfL, organized an AdCom for late March, and set a target action date of April 25, 2023.
Another example of success despite missing endpoints was achieved by Amylyx’ Relyvrio which was approved by the FDA in September 2022. The Phase II trial evaluated a combination of two molecules already in use[14] and achieved its primary measure of reduction in ALSFRS-R. However, it missed secondary endpoints including rate of decline in isometric muscle strength as measured by the Accurate Test of Limb Isometric Strength (ATLIS) device, the rate of decline in slow vital capacity (SVC) and survival among others. It was an uphill climb for Relyvrio, which required two AdComs and submission of additional data and analysis, nevertheless, the persistence paid off and approval was granted in both Canada and the United States.
What is the Correct Metric?
With so many promising candidates failing to achieve a statistically significant improvement in a primary endpoint, what is the best measure to use? There is extensive variability in patient preferences for an appropriate endpoint.[15] Despite its widespread use, 24-week ALSFRS-R may be too short to capture the benefits of effective therapies and in other cases the disease may be too advanced for the therapy to provide a measurable benefit.
Clene’s March Forward
With solid, but not perfect results, Clene plans future conversations with the FDA in an end of Phase II meeting to discuss the pathway forward. The unmet need in ALS is severe, and there is substantial interest in identifying therapies that improve important aspects of the patients’ condition. Additional data will also be generated in the open label extension (OLE) which is expected to produce actionable biomarkers that support CNM-Au8’s efficacy. The majority of those participating in the Healey trial elected to participate in the OLE, which may provide a rich trove of additional data.
Milestones
- Full data set from Healey trial – 1H:23
- ALS trial additional biomarker data – 1H:23
- Open label extension data – Summer 2023
- End of Phase II meeting with FDA – mid-year 2023
Summary
ALS has been in the public consciousness for decades despite its status as a rare disease. While early work on the disorder has been slow to produce a highly effective monotherapy, progress is being made both by sponsors of new candidates and in the regulatory space. In-development therapies may not be perfect but in the case of CNM-Au8, there is a favorable safety profile and signs of efficacy that may become more apparent when a longer-term view is taken. Survival was an especially positive endpoint for CNM-Au8 as two consecutive trials have demonstrated a 60% and 90% reduction in risk for this metric, respectively, in RESCUE-ALS (over 120 weeks) and Healey (24 weeks). Regulatory authorities are recognizing the difficult hurdles that ALS trials face in order to demonstrate efficacy and are evolving the standards to recognize this. One example is Amylyx’ Relyvrio which was approved by the FDA late last year. This suggests that the FDA is open to granting approval for candidates that can show a benefit and offer a benign safety profile.
As CNM-Au8 progresses through the ongoing study, consults with the FDA on next steps and presumably generates favorable biomarker results, we could see a pathway towards an accelerated approval followed by a confirmatory trial over the next year. In a rare disease with few effective treatments, the FDA has shown flexibility and evolution of its approach to approval. With the possibility of an NDA filing later this year, we believe Clene merits a closer look.
~~~~~~~~~~~~~~~~~~~~~~~~~~
DISCLOSURE: The author of this article has received compensation from the issuer directly, from an investment manager, or from an investor relations consulting firm, engaged by the issuer, for providing written content. John D. Vandermosten, CFA certifies that the views expressed in the research work accurately reflect his personal views about the subject security and issuers. No part of his compensation was, is, or will be directly or indirectly related to the specific recommendations or views contained in the research article.
[1] Source: Clene Inc. third quarter 2022 Form 10-Q.
[2] Nicotinamide adenine dinucleotide hydrogen
[3] Nicotinamide adenine dinucleotide
[4] Huang X, El-Sayed IH, Yi X, El-Sayed MA. Gold nanoparticles: catalyst for the oxidation of NADH to NAD(+) J. Photochem. Photobiol. B. 2005;81:76–83. Colloidal gold nanoparticles have previously been shown to catalyze the oxidation of NADH to NAD+ in a cell-free system.
[5] Source: Clene Corporate Presentation January 2023.
[6] The first group of participants in the Healey ALS Trial also included Biohaven Pharmaceuticals and RA Pharma.
[7] Source: Clene Presentation Slides, CNM-Au8 Topline Data, October 2022.
[8] When combined with placebo groups for the other therapies included in HAPT, the placebo group was comprised of 164 subjects.
[9] Source: Clene Presentation Slides, CNM-Au8 Topline Data, October 2022.
[10] The neurofilament light chain diagnostic uses blood samples to detect small quantities of cytoskeletal proteins that remain after the destruction of neurons.
[11] Urinary p75ECD, or in longer form, urinary neurotrophin receptor p75 extracellular domain is a prognostic, disease progression and pharmacodynamic biomarker and has demonstrated a correlation with ALS diagnosis.
[12] Elevated levels of ubiquitin C‐terminal hydrolase L1 (UCHL1) are present in ALS patients and may provide a biomarker for early diagnosis.
[13] Van Eijk, R.P.A et al. Composite endpoint for ALS clinical trials based on patient preference: Patient-Ranked Order of Function (PROOF). J Neurol Neurosurg Psychiatry. 2022 May; 93(5): 539–546.
[14] The two compounds are sodium phenylbutyrate and tauroursodeoxycholic acid.
[15] Van Eijk, R.P.A., et al. Composite endpoint for ALS clinical trials based on patient preference: Patient-Ranked Order of Function (PROOF). J Neurol Neurosurg Psychiatry. 2022 May; 93(5): 539–546.