Poster Session at HFSA Sept. 13th and 14th Philadelphia and Oral Presentation 4pm Sept. 28th at TCT Cardiology San Francisco
Second Heart Assist, Inc., developer of a percutaneous placed circulatory assist support pump within an aortic stent, announced that data from a first in human study and summary of four years of prior pre-clinical studies will be presented at both the Heart Failure Society of America (HFSA) and the Transcatheter Cardiovascular Therapeutics (TCT) Conference in San Francisco this month.
The results of the first in human pilot study and a brief summary of 4 years of pre-clinical testing will be presented on the following dates and times:
* Sept. 13th - Second Heart Assist Circulatory Assist Pump with an Aortic Stent First in Human Experience - Late Breaking Clinical Trials section 6 to 7:30pm # LBCT - 012 in the Exhibit Hall at HFSA’s 23rd Annual Scientific Meeting Philadelphia, PA.
* Sept. 14th - Second Heart Assist Circulatory Assist Pump with an Aortic Stent First in Human Experience - Late Breaking Clinical Trials section 6 to 7:30pm # LBCT - 012 in the Exhibit Hall at HFSA’s 23rd Annual Scientific Meeting Philadelphia, PA.
HFSA 2019 is at the Pennsylvania Convention Center and Philadelphia Marriott Downtown, 1201 Market Street Guest Entrance, 1200 Filbert St At, Philadelphia, PA 19107 Sept. 13th to 16th, 2019.
* Sept. 28th 4pm - Improving Renal Perfusion Pressure with Second Heart Assist Circulatory Assist Pump within an Aortic Stent - @ TCT Cardiology Annual Conference, Moscone Convention Center, Room 156/158, 747 Howard St, San Francisco, CA 94103 - INTERVENTIONAL HEART FAILURE AND HEMODYNAMIC SUPPORT: CHRONIC THERAPIES SESSSION - https://www.crf.org/tct/program/agenda/view-full-agenda
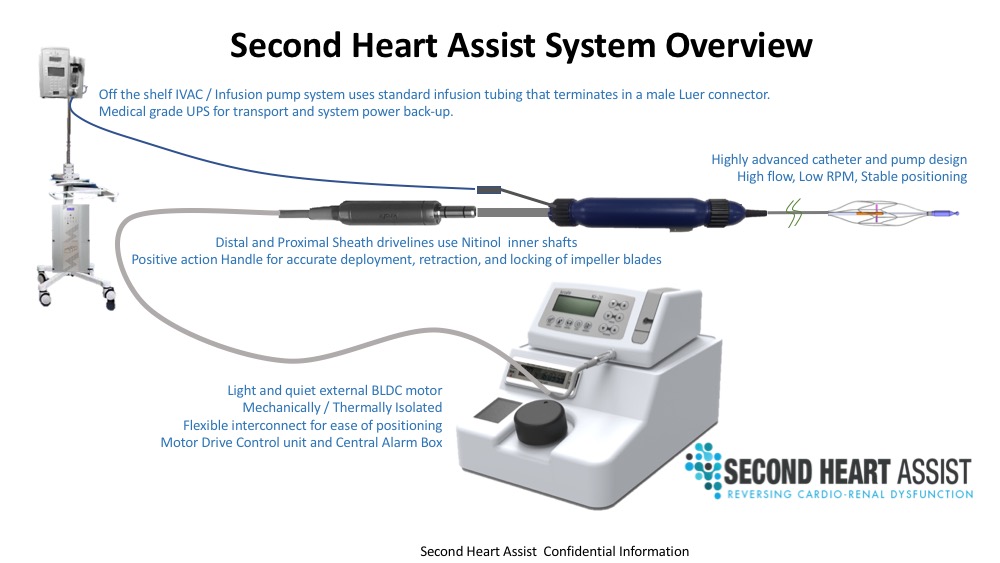
About Second Heart Assist, Inc.:
Focused on development of a circulatory assist pump within an aortic stent with R&D and Product Development labs in Utah and Los Angeles with affiliated product development and testing labs in Minneapolis, Paraguay, Brazil, Pittsburgh, Australia and Canada. Products are not yet proven to be safe or effective and are available for investigational use only in countries where proper regulatory clearances have been granted and hospital IRB ethics committee approval has been received along with an appropriate patient consent process. Not available in the USA at this time. Prior to completing the first in human study August of 2019 the Second Heart Assist team completed multiple large animal studies in Boston, Columbus and Minneapolis, mock loop studies in the USA in Louisville, Minneapolis, MN and in Victoria, BC, Canada, multiple rounds of durability challenge testing up to 9 days and multiple rounds of computational fluid dynamics assessments in Germany and the USA. See www.secondheartinc.com for more information.























